Clinical trials are what guarantee pharmacological safety and the improvement in the method allows drugs and therapies to be developed today with the highest ethical, scientific and quality guarantees.
But there was a “first clinical trial”, the cornerstone, the origin of everything; a history of sailors and diseases that dates back to 1746.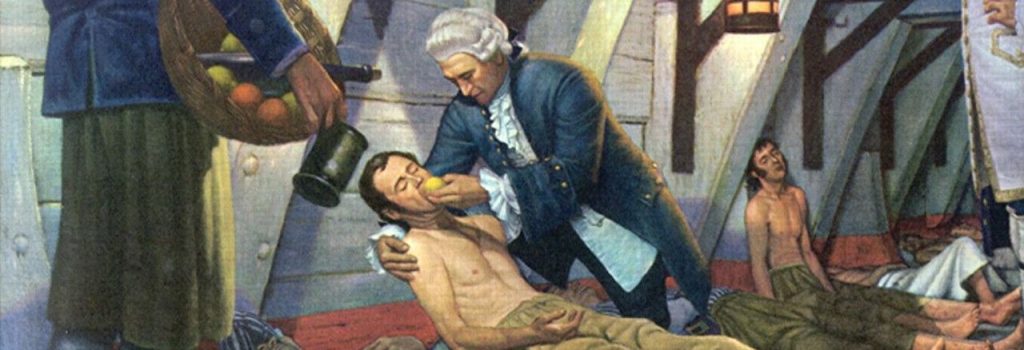
Scurvy led James Lind to conduct the first clinical trial..
At that time, scurvy – a disease caused by a lack of vitamin C or ascorbic acid – was developing at an alarming rate among sailors who suffered severe gum lesions, skin sores, became prostrate and died.
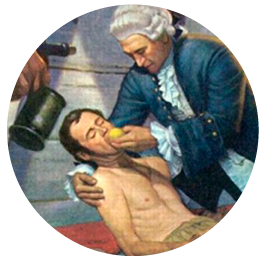
On one of those long voyages, James Lind – a Scotsman now considered the father of naval medicine – divided 12 sick sailors into six pairs and each pair was given a different supplement to their diet: cider, vitriolic elixir (diluted sulfuric acid), vinegar, seawater, two oranges and a lemon, or a purgative mixture.
As a result of what has been considered “the first clinical trial in history”, only the two sailors who took the fruit improved. Thus it was empirically proven that citrus fruits prevented scurvy. With this act, Lind initiated the way to modern medicine as we know it today.
The success: the control of variables: Lind chose patients with similar symptoms, kept them in the same place and provided them with a common diet.
As a term, clinical trial appeared for the first time in an anonymously published journal called Lancet in 1931, coinciding with the constitution of the UK Clinical Trials Committee. A few years later Sir Austin Bradford Hill, today considered the father of the modern clinical trial, published the first book on the subject: “Principles of Medical Statistics”.

Clinical trials are the basis of scientific research.
The first controlled clinical trial dates back to 1946; just two years after the discovery of streptomycin to test the efficacy of this drug in the treatment of tuberculosis.
276 years after that first trial, clinical research continues to evolve constantly and clinical trials represent the possibility of access to new drugs or treatments, essential for access to a better quality of life and, in many cases, for survival.
Highly regulated, clinical research is governed by the regulatory bodies of each country, but also by international standards, global good clinical practice and global and local ethics committees.
The role of those 16th century sailors is similar to that of VOLUNTEERS today: those billions of people around the world who enable billions of others to benefit from their generosity.
According to the WHO, childhood vaccines save the lives of four million children a year by providing protection against diseases such as diphtheria, measles, pneumonia, rotavirus, rubella, tetanus and polio.

According to the WHO, childhood vaccines save the lives of four million children a year by providing protection against diseases such as diphtheria, measles, pneumonia, rotavirus, rubella, tetanus and polio.
Clinical trials are what guarantee pharmacological safety and the improvement in the method allows drugs and therapies to be developed today with the highest ethical, scientific and quality guarantees.

Clinical trials are what guarantee pharmacological safety and the improvement in the method allows drugs and therapies to be developed today with the highest ethical, scientific and quality guarantees.

Days like today are what we work hard for, where a small victory means a big step for the community, especially for those suffering from diseases and for their families and friends who accompany them.

Days like today are what we work hard for, where a small victory means a big step for the community, especially for those suffering from diseases and for their families and friends who accompany them.

It is a commonly accepted myth that people are used as guinea pigs in medical experiments. In reality, this myth is false and no information has been found to validate this assumption.

It is a commonly accepted myth that people are used as guinea pigs in medical experiments. In reality, this myth is false and no information has been found to validate this assumption.
Clinical trials play a crucial role in the fight against cancer. These studies are fundamental to evaluate the effectiveness and safety of new treatments and therapies, as well as to improve cancer care in general.

Clinical trials play a crucial role in the fight against cancer. These studies are fundamental to evaluate the effectiveness and safety of new treatments and therapies, as well as to improve cancer care in general.

Based on the premise that medical research is the only tool that Science has to study new and better treatments for diseases, from BRCR – as a key link – we are committed to the task of providing the quantity and quality of information necessary for the peace of mind and safety of all the actors involved in the processes.

Based on the premise that medical research is the only tool that Science has to study new and better treatments for diseases, from BRCR – as a key link – we are committed to the task of providing the quantity and quality of information necessary for the peace of mind and safety of all the actors involved in the processes.
In the world of medicine, each new drug or treatment that reaches our hands has gone through a rigorous series of tests and stages before being approved for marketing.

In the world of medicine, each new drug or treatment that reaches our hands has gone through a rigorous series of tests and stages before being approved for marketing.
8200 W Sunrise Blvd – Suite D2
FL 33322
Phone: +1 888 745 2727
Ashford Medical Center
29 Washington Street suite 310
San Juan, PR 00907
Phone +561 447 0614 Ext. 504
Sur 132 No. 108 Suite 601-605 Col. Las Américas CP 01120
Phone: + 52 55 2614 5118
Contact our Headquarter
Phone +1 888 745 2727